An ambitious EU Trade Policy focused on Jobs and Growth
As Pfizer, we serve patients in over 170 countries and have a strong presence across the world, including in the European Union (EU). As such, we depend on an open, predictable, rules-based global trading environment to ensure that patients around the world can access our medicines.
EU Trade Policy supports innovation and access to medicines

Global trade is a major driver for jobs and growth in Europe. Nowhere is this more evident than in the innovative pharmaceutical sector. Despite an often challenging environment in Europe, and increasingly protectionist policies outside of Europe, exports of medicines have been a source of economic resilience1.
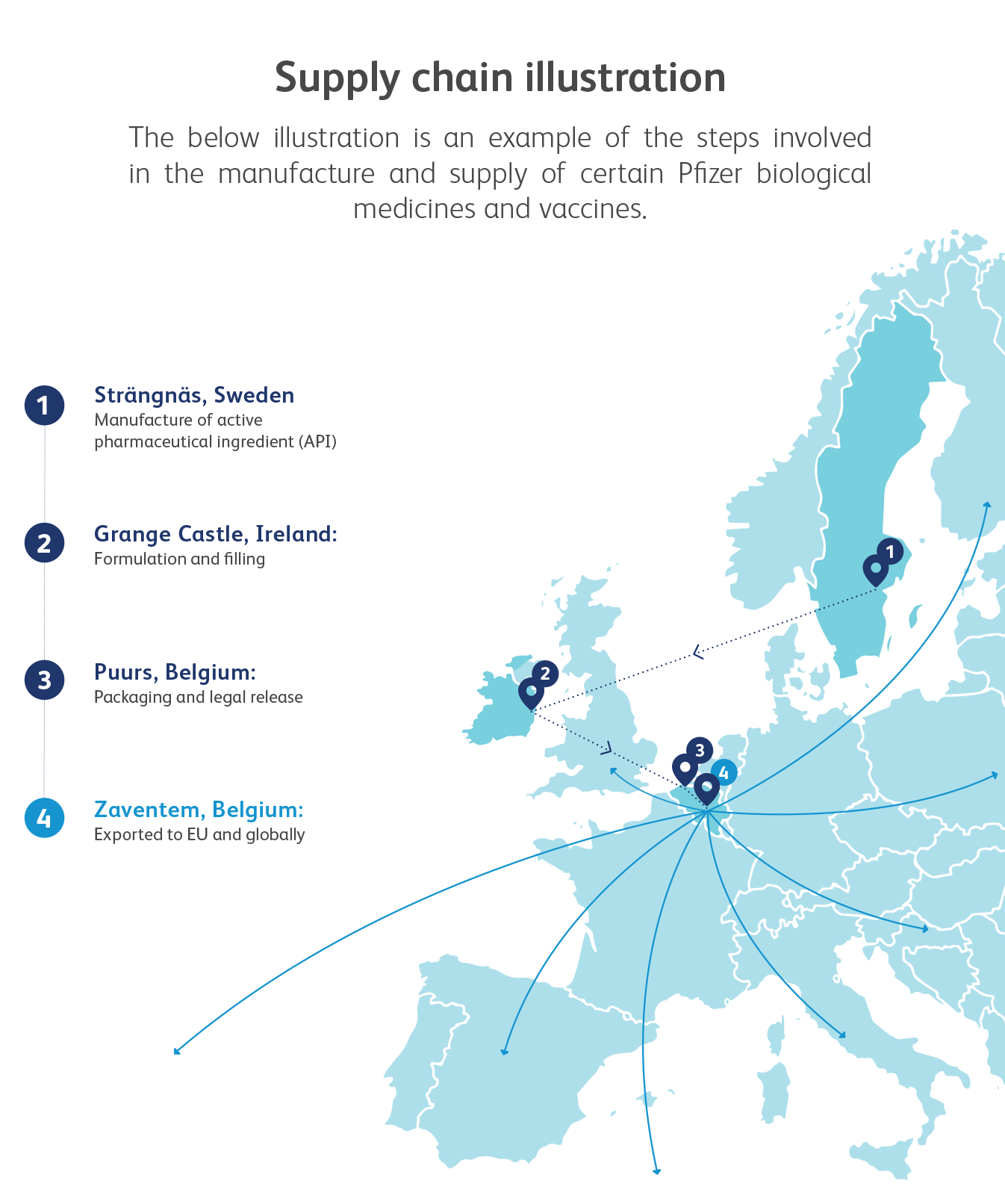
Pfizer alone employs thousands of people who research, develop and manufacture medicines in sites across Europe, and export them around the world2.

This work is underpinned by the EU’s external trade policies, on areas such as removal of protectionist barriers, safeguarding of effective regulations, and fostering effective intellectual property (IP) protections. Taken together, these help to create an environment for continued investment in healthcare innovation, and to ensure access to medicines for patients.
Free trade agreements (FTAs) provide a win-win by removing trade barriers, helping to make medicines available to a greater number of patients, and increasing global competitiveness.
At Pfizer we believe that IP rights are critical to foster innovation and provide incentives for future R&D investment. We support efforts to strengthen IP rights and enforcement in FTAs negotiated by the European Union, the United States and other countries.
Our sector’s ability to operate in the EU is underpinned by a world-leading, science-based regulatory framework. Internationally, the EU plays acritical role in advancing both its own, and globally-recognised, standards (e.g. WHO), raising the bar globally.

Key policy challenges and opportunities
At a time of major uncertainty and increasing protectionism, the effectiveness of institutions such as the World Trade Organisation (WTO) is crucial to defending the bedrock of our economy.
As countries negotiate new traderules, it is equally important that they continue to implement and enforce existing rules. Effective enforcement is necessary to ensure the economic benefits of free trade for all members of the global trading system
Despite differences of approach on trade policy, relations with the United States are built on deep economic foundations and shared values. Opportunities exist to further the strong cooperation between world-leading regulators on both sides of the Atlantic, and to work together where possible to address shared policy concerns at a global level.
We live in a crowded and competitive global environment: improving the ability to export to, invest in, and trade with growing markets is hugely important.
Trade policies should reflect the evolving nature of technology (including digital), the economy and the broader world around us. This can support patient access to innovative, safe and effective medicines around the world.
Key Policy Points
During the 2019-2024 legislature, we call on the EU and its Member States to:
- Continue to defend the rules-based global trading system;
- Secure as close and positive as possible a future relationship with the United Kingdom (UK), notably one that would allow for close alignment in the regulation of pharmaceuticals;
- Build on the depth of existing ties with the United States;
- Ensure the EU’s trade agenda for the coming years seizes major opportunities, particularly in the Asia-Pacific region;
- Not only maintain Europe’s own high IP standards, but continue to advocate for effective IP globally, enabling innovation to flourish.
References
1 European Federation of Pharmaceutical Industry and Associations (EFPIA), The Pharmaceutical Industry in Figures, 2018, p. 3.
2 For more information, see the accompanying paper “Pfizer in Europe – Facts and Figures”, also available at www.pfizereupolicy.eu.