How do we develop medicines?
Developing a medicine takes many years and major investment. Regulatory authorities approve a medicine based on extensive data on its quality, safety and efficacy for the condition it is intended to treat. Generating the data to meet these requirements defines the process for medicines development, meaning that the EU’s regulation on medicines is highly influential. At Pfizer, we are committed to develop breakthroughs that change patients’ lives.
Pre-clinical research
Every medicine works for a specific chemical and biological reason, a so-called ‘mechanism of action’ (MoA). Based on advances in basic scientific understanding, scientists explore medicines candidates according to a specific MoA.
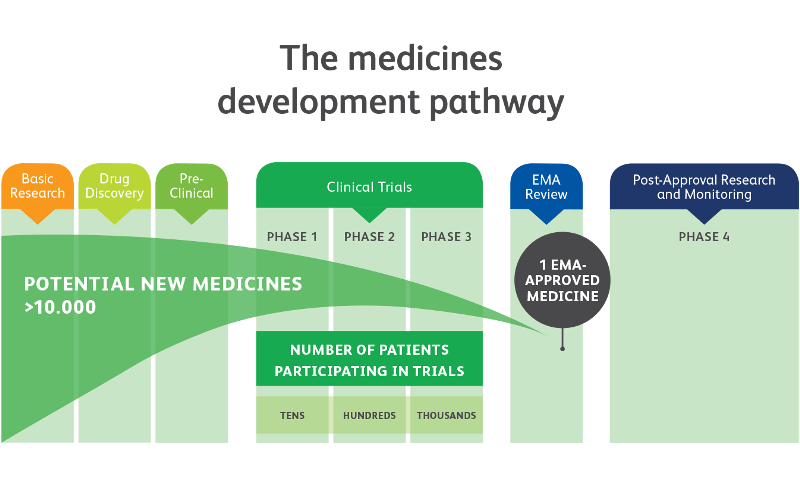
Pre-clinical research identifies candidates that may be effective and safe. Typically thousands of molecules are examined for every one that makes it into a clinical trial.
Clinical research1
Clinical trials start with testing in a small number of people in Phase 1, which is designed to provide early indications about the safety of the new drug.
If all the requirements of Phase 1 are met, the drug progresses to Phase 2 trials, where safety and efficacy are tested, generally in a slightly larger number of target patients.
Promising drugs that achieve their Phase 2 goals are then usually tested in larger numbers of patients in Phase 3 trials, which are designed to generate data on efficacy and safety that can support an application for regulatory approval. Approval is obtained on the basis of an assessment of the potential benefits and risks to patients.
Following approval, Phase 4 trials often continue to generate data to further increase understanding of the performance of the medicine. If a medicine goes on to demonstrate an unacceptable level of risks for the benefits it provides, regulatory authorities can still remove the medicine’s license at this stage.
Developing a medicine in practice
Developing a medicine that meets the rigorous requirements of regulatory agencies is costly and often takes more than a decade. When taking failures into account, the average cost of developing a medicine has been estimated at €2.57bn2.
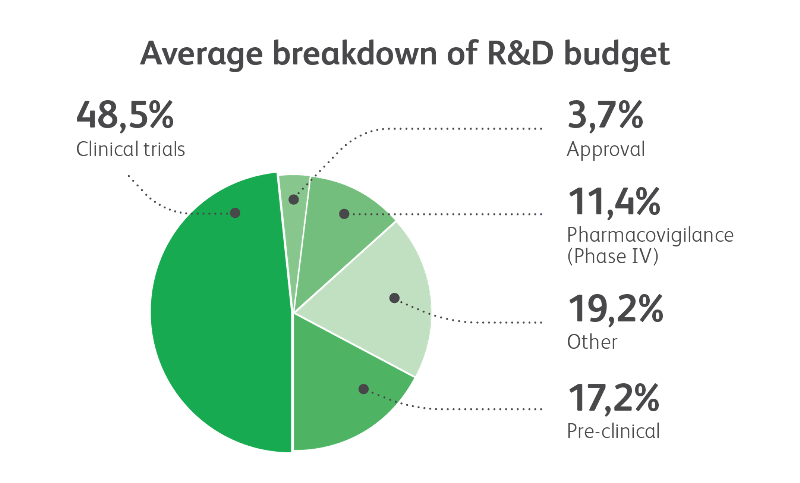
Pharmaceutical R&D is not restricted to those places with pre-clinical laboratories: clinical trials represent roughly half of development costs, a major investment in research in every Member State.
Some medicines are developed entirely within a single pharmaceutical company, while others may be developed through partnerships, in-licensing and acquisitions. The complexity of progressing a medicine through the development process is a major factor in smaller companies choosing to work with larger companies to reach approval.
Medicines for rare diseases and for children are developed in specific regulatory and intellectual property frameworks4.
Patient involvement in R&D
As the goal of any medicine is to improve patients’ lives, it is imperative to obtain patient insights to help shape drug development. Pfizer is committed to increasing the inputof patients and patient representatives during the drugdevelopment process.
Pfizer supports the European Patients Forum (EPF)’s EUPATI project, which helps to train patients on medicines development.5 We also engage in relevant public-private research projects, aimed at ensuring patient involvement in development, as part of the Innovative Medicines Initiative (IMI).6
The roles of public and private research
The public and private sectors play complementary roles in health research and medicines development. The former is more focused on basic research of disease progress and mechanisms; the latter is more focused on the discovery, synthesis and testing of compounds based on that knowledge.
Given the high rate of failure in drug development, pharmaceutical companies take considerable risk in investing their capital in this process. This level of cost and uncertainty generally prevents public investment in the later stages of medicines development. The innovative pharmaceutical industry’s R&D investment in Europe in 2017 amounted to more than €35 bn.7
Key Policy Points
During the 2019-2024 legislature, we call on the EU and its Member States to:
- Take a strategic approach to supporting biomedical R&D that can make the EU a world leader in this area, as outlined in the EFPIA 2019 manifesto8, through e.g. research funding, regulatory requirements, and predictable single market conditions;
- Maintain a stable and competitive intellectual property (IP) framework, essential in a sector that often invests for over a decade before its products reach the people who need them;
- Ensure a world class regulatory system, able to accommodate emerging science while providing predictability for development programmes, and fast-track breakthrough therapies.
References
1 See Figure 1 for an overview of the medicines development process.
2 J. A.Di Masi, H. G. Grabowski, R. W. Hansen, “Innovation in the pharmaceutical industry: New estimates of R&D cost”, Journal of Health Economics, Vol. 47, May 2016, pp 20-33.
3 European Federation of Pharmaceutical Industry and Associations (EFPIA), The Pharmaceutical Industry in Figures,2018.p.8. See Figure 2 below for a breakdown of R&D costs.
4 For more information, read our fact sheet on ‘How does pharmaceutical IP work?’, also available on www.pfizereupolicy.eu
5 European Patients’ Academy on Therapeutic Innovation (EUPATI).
6 Patients Active in Research and Dialogue for an Improved Generation of Medicines (PARADIGM).
7 EFPIA, The Pharmaceutical Industry in Figures, 2018, p.7.
8 EFPIA, Building a Healthier Future for Europe, Brussels, 2019.